Left or right?… Distinguishing chiral nanoparticles by their strongly asymmetric photoemission yield
Many biological and bio-active molecules, like pharmaceuticals, fragrances exist in two distinct mirrored forms, the enantiomers of a so-called chiral species. While identical in chemical and physical properties, two enantiomers of the same molecules differ in their interaction with a given chiral environment, making the challenging analytical distinction of enantiomers a crucial task in industry.
Research conducted on the DESIRS beamline has now revealed a pathway to a simple table-top experimental approach to identify enantiomers of a molecule and quantify their relative content in condensed sample particles.
Enantiomers of chiral molecules, like our hands, are mirror images one of the other but cannot be superimposed. Such chiral molecules are omnipresent in biology, biochemistry, and pharmacology, where analytical enantio-sensitive measurements are crucial. A tragically famous example of the importance of this enantio-sensitivity is the drug known as Thalidomide: the left-hand form of Thalidomide molecule is a sedative, while the right-hand form has teratogenic effects; a mixture of the two forms was prescribed as a drug for pregnant women in the 1960s, resulting in the birth of thousands of children with birth defects. However, most traditional chiroptical techniques provide only weak chiral signatures, making precise measurements difficult.
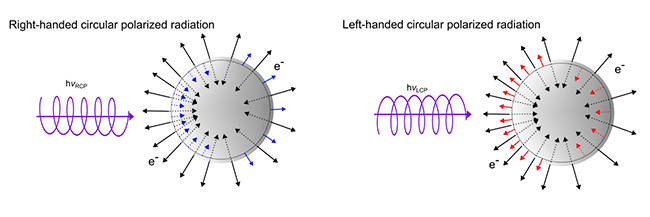
Photoelectron circular dichroism (PECD1) offers orders of magnitude more intense chiral asymmetries. It occurs when circularly polarized light ejects electrons from a given enantiomer, producing an asymmetric angular distribution of electrons depending on the molecule’s handedness, see figure 1. Until now, this effect has mainly been studied by carefully measuring the direction of emitted electrons using complex instruments and high vacuum environments, limiting the technique to relatively small, stable molecules excluding many molecules relevant to biology and pharmacology.
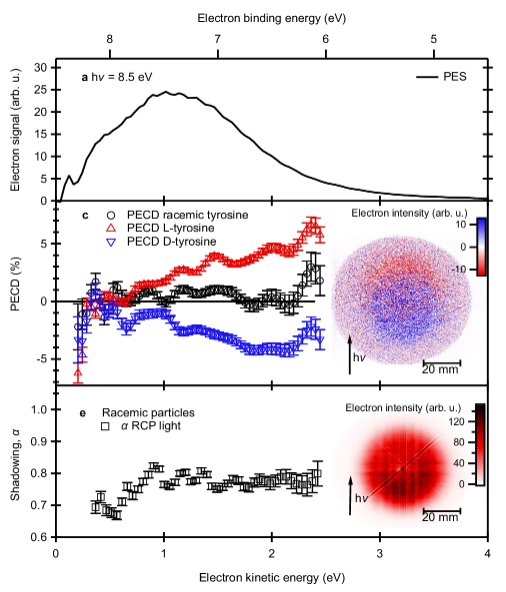
The key advance of this work is showing that this directional asymmetry can also be detected in a much simpler way: by measuring the total number of emitted electrons from a condensed sample particle. The researchers from the DESIRS beamline of synchrotron SOLEIL, the Vinča Institute of Nuclear Sciences at the University of Belgrade, Serbia and the Institute of Physics of the University of Freiburg, Germany demonstrate that, in small nanoparticles of the enatio-pure amino acid tyrosine suspended in air (aerosols), PECD translates into a measurable difference in overall photoemission yield, i.e. how many electrons are emitted in total.
This happens because of a combination of two effects. First, PECD creates an intrinsic asymmetry in how electrons are emitted inside the particle. Second, a “shadowing” effect, caused by how light penetrates and is absorbed within the nanoparticle, induces another, non-chiral, asymmetry in the forward/backward emitted electrons. This second effect changes which electrons can likely escape from the particles. Together, these effects convert directional asymmetry into a detectable difference in total electron emission yield, see figure 2.
Experimentally, the team studied the shadowing and PECD effects on nanoparticles of about 100 nanometers in size using VUV synchrotron radiation and photoelectron imaging at the DESIRS beamline. Similar particles had previously shown remarkable chiral asymmetries in the photoemission yield, that have not been linked to the PECD effect. The authors of the current study used theoretical models to simulate how light interacts with the particles and how electrons escape from their surfaces. The results show that the combined PECD & shadowing effects can produce surprisingly large asymmetries in electron yields, on the order of several percent to around 10 %, typical for PECD but much stronger than conventional chiroptical signals.
A major implication is practical: instead of relying on sophisticated electron imaging detectors and vacuum systems, chirality could potentially be measured using simpler setups that only track total electron yields produced by a simple UV photon source. This opens the door to more accessible and sensitive analysis of chiral materials, including biological molecules that are currently challenging to study with PECD techniques, atmospheric aerosols, or nanoparticles used in medicine and catalysis.
In summary, the study establishes a clear link between a detailed microscopic effect (PECD) and an easily measurable macroscopic signal (total photoemission yield). By doing so, it provides a new and promising approach for detecting and quantifying molecular chirality in complex, real-world particulate systems.
1- PhotoElectron Circular Dichroism (PECD): As Circularly Polarized Light (CPL) is a chiral object, it will induce enantio-specific processes upon interaction with a given enantiomer of a chiral species: these are circular dichroisms, a chiral recognition between chiral objects, such as a left glove "recognizes" a left hand. In the case of photoemission of a given enantiomer of a chiral molecule by an CPL of a given helicity (left or right), the PECD manifests itself by a forward / backward asymmetry with respect to the light propagation axis, which is not found for non-chiral species. This asymmetry is reversed when the enantiomer or light helicity is swapped. The specificity of PECD, as compared to other types of circular dichroisms, lies in its intensity: from a few % to a few tens of %. It is also very sensitive to molecular structures such as isomers (molecules with the same atomic composition, but a different arrangement of these atoms), or conformers.
