Tailor-made self-assembly of nanostructures to produce electronic components with controlled characteristics
From next-generation displays for stretchable and bendable cellphones to electronic tattoos that can measure biometrics for improved health, the understanding of conjugated polymer* self-assembly is critical. The study described here reports the nanoscale assembly of poly(3-hexythiophene) (P3HT), a common conjugated polymer, which can be driven and stabilized by oxidative doping. Results were confirmed using GIWAXS at SIRIUS beamline.
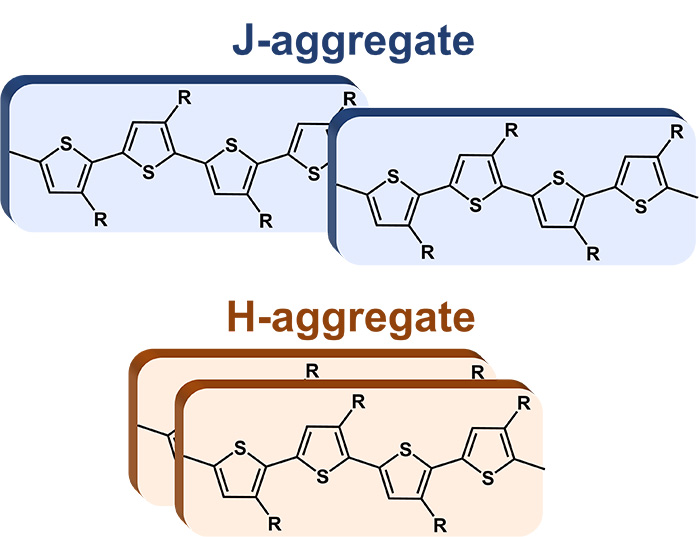
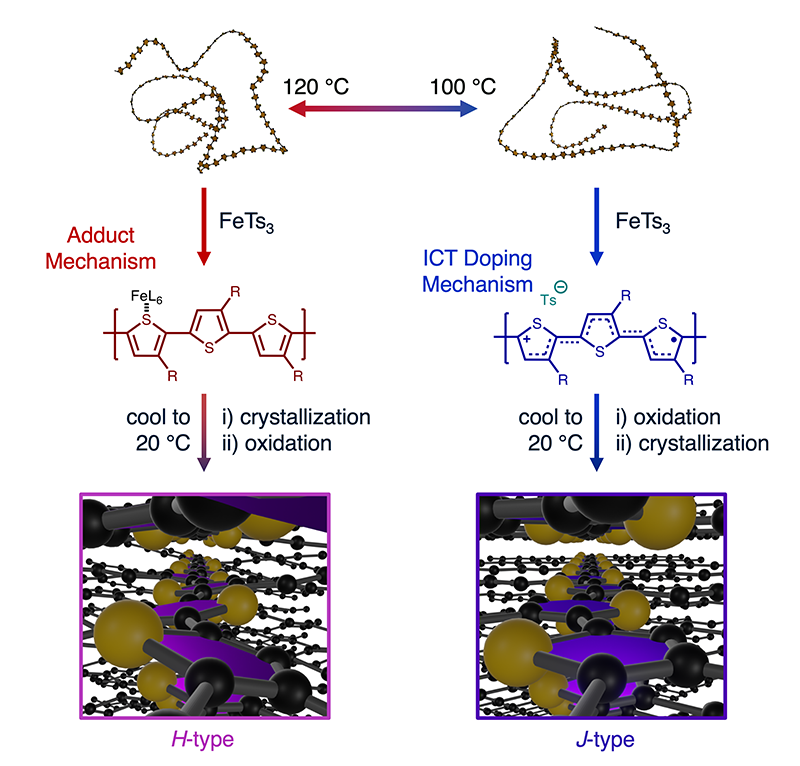
Researchers at the Universities of Toronto and Ottawa have developed a simple, robust and accessible method for preparing organic electronic components based on P3HT, an electrically conductive polymer. These components aggregate into multi-micrometers nanofiber bundles or form nanoparticles of 20-40 nm depending on their state of aggregation (J/H, see Figure 1). In order to improve their conductivity, these materials have to be doped, usually by the addition of Iron III. Both the electronic states, the colloidal stability and the self-assembly of the polymer nanostructures can be adjusted by the conditions and level of such oxidative doping (concentration, temperature).
The method described here is the first for simultaneously controlling the oxidative doping level, H/J aggregate character, morphology, and nano/micro-structure size over such a wide range of length scales. The nanostructure formation mechanism depends fundamentally on the polymer conformation in solution; temperature is only one of many factors that govern polymer conformation.
GIWAXS measurements on SIRIUS
At SIRIUS beamline, Grazing-Incidence Wide-Angle X-ray Scattering (GIWAXS) was performed on oxidized nanostructures deposited on glass slides. Samples were prepared by pipetting 300 microliters of the oxidized nanostructure solution onto a clean glass slide and then removing the solvent in-vacuo to form a thin layer of nanostructures and inhibit any additional ordering during the deposition process.
GIWAXS measurements were performed on oxidized nanostructures prepared at several temperatures (T = 100, 110, and 120 °C) with 5, 10, 15, 20 % FeTs3 (dopant), spanning the full domain of J-aggregate values.
The analysis of the GIWAXS measurements allows to trace the adopted configuration and crystal parameters of the oxidized P3HT nanostructures. The orientation adopted during the deposition can thus be determined: either edge-on or face-on. It was also possible to deduce that, depending on the doping conditions (temperature and FeTs3 concentration), either purely H nanostructures or H/J mixtures are obtained. Furthermore, it appears that the edge-on configuration is the only one adopted by the J-type oxidized structures, whose ordering is therefore higher and more uniform than that of the H-type structures.
Finally, the quantitative analysis of the variation of the crystal parameters allows to understand the chemical mechanisms involved in the doping. Thus, the GIWAXS results make it possible to characterize the self-assembly of the aggregates that have formed as a function of temperature and dopant concentration and to understand how these aggregates order and orientate themselves. This information is essential to determine how the material will behave in an electrical device where it is crucial to be able to control the self-assembly.
Thus, this study shows that by controlling the doping process, it is possible to control the aggregate so that it follows the intended self-assembly pathway. GIWAXS helps to determine which method is most promising.
*conjugated polymer: after certain modifications, a polymer can become electrically conductive. To do this, the polymer must be conjugated, i.e., the polymer's main chain must have alternating single and multiple bonds, and it must be "doped", which means that electrons are removed (by oxidation) or added (by reduction).